IF YOU'RE HERE BECAUSE you’re revising for your school biology exams but didn’t understand your teacher’s explanations… this page isn’t quite intended for you, but welcome anyway! I hope you find it useful.
If you’re here because you’re trying to get pregnant, you’re definitely in the right place. Once you’ve finished reading, you’ll have a far better understanding of your embryos, how chromosomal defects occur, who’s at risk, and lots more.
Contents
Why the fuss about chromosomes?
Chromosomal issues are one of the leading causes of implantation failure, miscarriage and birth defects among babies. Some people are more at risk of producing embryos with chromosomal issues than others.
There are two types of screening that intend to discover if any chromosomal issues exist in your embryos: PGS (preimplantation genetic screening) and PGD (preimplantation genetic diagnosis). This article isn't about PGS or PGD, though: instead, it explains why and how chromosomal issues appear in the first place.
Think of this article as containing all the background information you need to fully understand PGS and PGD (rather than “essential reading”).
- If you’d rather just learn about the PGS process, how it works, the success rates, pros and cons, etc., read this article about PGS instead.
- I haven’t yet written an article about PGD, but it’s on the to-do list!
What is a chromosome?
A chromosome is a small, dense strand of DNA, which contains genes that tell your body how to develop and function. (Chromosomes also contain proteins, which will become relevant later.)
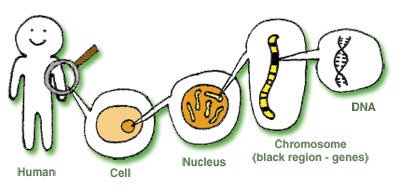
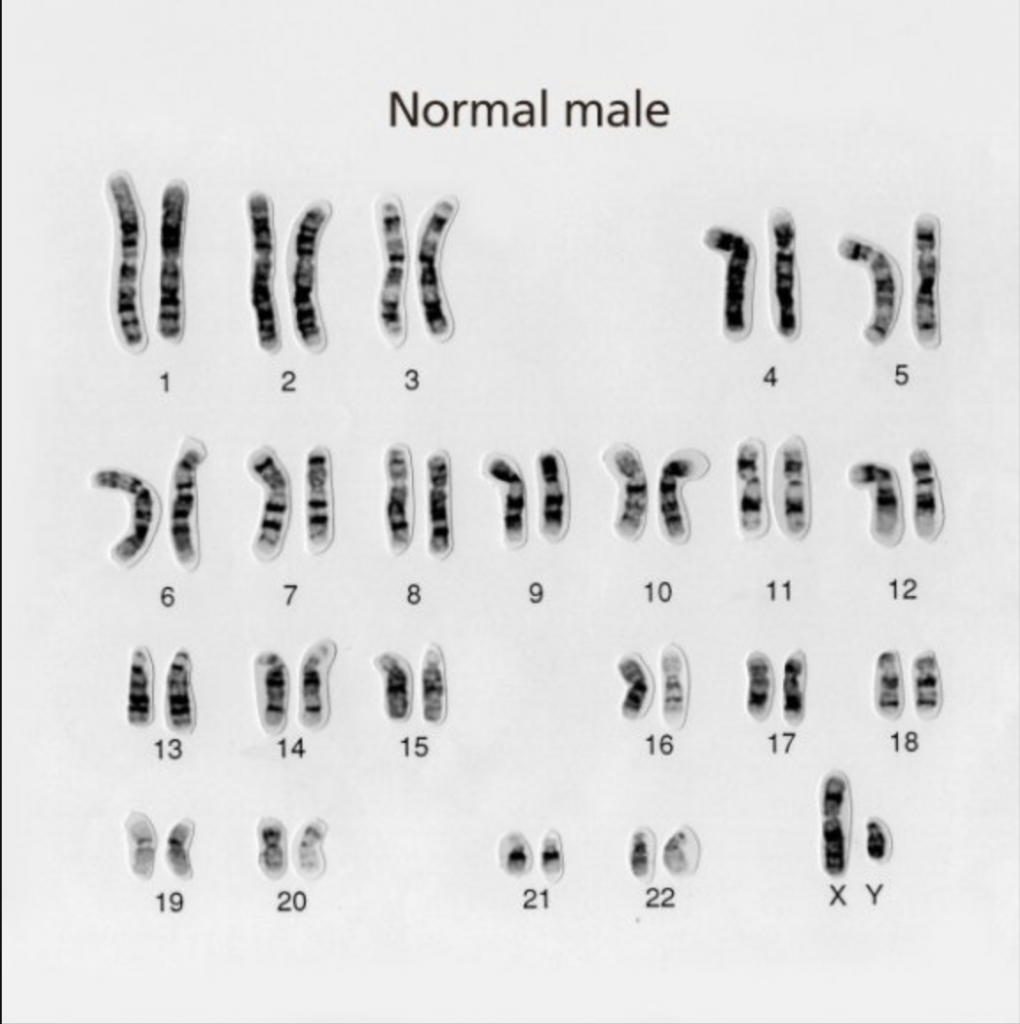
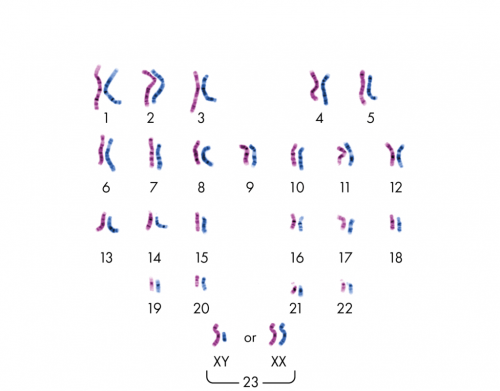
Every cell in the human body should contain 23 pairs of chromosomes – making 46 chromosomes in total. A set of 46 chromosomes, as seen under a microscope, is known as a “karyotype”. Like this:
Some background info on these 23 pairs of chromosomes…
So… every cell in the human body should contain 23 pairs of chromosomes – making 46 chromosomes in total.
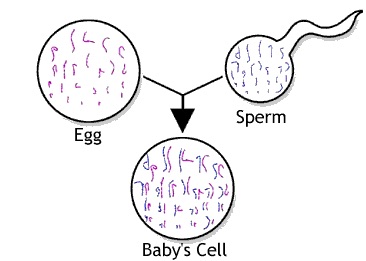
Every cell except egg and sperm cells, which contain just one half of the set each. That is, the egg contains 23 chromosomes and the sperm contains 23 chromosomes.
When an egg and sperm fuse during fertilisation, the chromosomes from the egg and sperm pair up into matched sets known as “homologous” pairs (with the slight exception of the final, 23rd pair, which we’ll come to shortly).
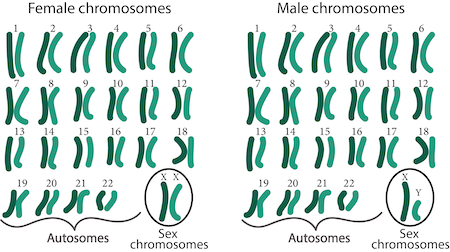
The first 22 pairs of chromosomes are called “autosomes”
These autosomes are numbered from 1 to 22 (from largest to smallest). The autosomes contain most of your genetic information, such as height, eye colour, and so on.
“Homologous” means that the chromosomes in each pair share the same size and shape, and carry the same type of genetic information – but it doesn’t mean they’re identical. Even though homologous chromosomes carry the same type of genetic information, they don’t necessarily have the same versions of genes. That’s because one of those chromosomes came from the mother, while the other came from the father.
Here’s an example to show what I mean:
There’s a gene on Chromosome pair #9 that determines blood type. It’s possible for a person to have two identical copies of the gene – one on each homologous chromosome. E.g. a person may have the “Type A blood” gene from both the mother and father. But a person could have two different gene versions – such as one for “Type A blood” and one for “Type B blood”.
The final, 23rd pair is called the “sex chromosomes”
And it determines a person’s shoe size.
Kidding! The sex chromosomes determine a person’s sex.
The 23rd chromosome of the egg is always referred to as an “X” chromosome, but the 23rd chromosome of the sperm can be an “X” or a “Y”. When the egg and sperm fuse during fertilisation, the resulting 23rd pair can either be “XX” or “XY”, depending on which chromosome the sperm is carrying. An “XX” pair indicates that the embryo is female, and an “XY” pair indicates that it’s male.
If you squint a bit and blink a bit and hold one eye shut, the “X” sort of looks like an X and the “Y” sort of looks like a Y. You might need a few glasses of wine to really see it:
All about chromosomal abnormalities
What is a chromosomal abnormality?
Any deviation from the normal karyotype is known as a chromosomal abnormality; at least 50% of all miscarriages are due to a chromosomal abnormality. There are two main types of deviation:
- Numerical abnormalities – where the number of chromosomes in each cell is anything other than 46 (or 23 pairs). Numerical abnormalities are far more prevalent than structural ones, and they account for about 95% of the miscarriages that are caused by chromosomal abnormalities. Only about 0.3% of babies are born with a numerical abnormality, because most are miscarried.
When embryos have the incorrect number of chromosomes, they’re called “aneuploid”. When they have the correct number, they’re called “euploid”. - Structural abnormalities – where large sections of DNA are either missing from or added to a chromosome. They account for the remaining 5% of miscarriages that are caused by chromosomal abnormalities. (But unlike numerical abnormalities, it’s far more likely that babies can survive with structural abnormalities – and some are completely harmless.)
How common are chromosomal abnormalities in embryos?
Frighteningly common. According to one study, 42.9% of embryos (across all age ranges) are found to be aneuploid.
But chromosomal abnormalities increase dramatically with maternal age. Here’s some data from a study conducted in 2012 (note that the second column shows the percentage of normal embryos):
| Maternal age | % normal embryos |
| < 25 years | 70% |
| 25–29 years | 48% |
| 30–34 years | 42% |
| 35–39 years | 34% |
| > 39 years | 11% |
Igenomix (a reproductive genetics company) had similar results:
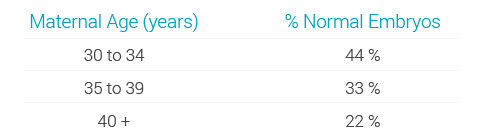
And this study from 2014 found that women under the age of 35 had a 60% chance of a blastocyst (the name for an embryo five or six days after fertilisation) being euploid, which reduced to 30% by the age of 41. The chance of getting NO euploids was about 10% for under 35s and about 50% by the age of 43. Other studies (like this one, but many others too) have the same sorts of figures.
Why do abnormalities become more common as women get older? We’ll cover that in a LOT of detail later!
If one cell in a human body contains chromosomal abnormalities, does that mean all cells in that body contain chromosomal abnormalities?
For the purposes of the discussion right now, we’ll say “yes, usually”. But if you want the full, nuanced answer, read my article about PGS testing (read the bit about “mosaicism”).
Here’s why:
- When a sperm fertilises an egg, a single cell called a zygote is formed. The zygote contains all 46 chromosomes (23 from the egg and 23 from the sperm).
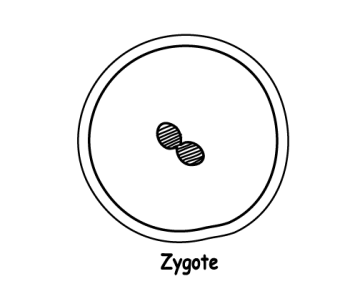
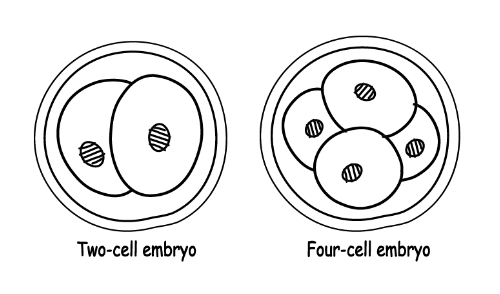
- The new cell divides in two for the first time after 24 hours and it’s no longer called a zygote: it’s now an embryo. A short while later, it divides in two yet again. (If you want to do more Googling about this process later, it’s called “mitosis”.)
- This cell division keeps happening until you end up with so many cells that they can’t be counted.
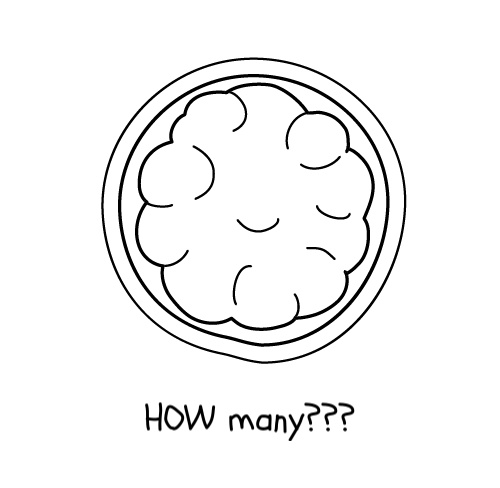
- And now for the important bit: as the embryo grows and the cells divide, the chromosomes are copied in every single cell. All the cells in a human body are made this way, which means every cell has an identical set of chromosomes. An error in the first cell, therefore, will be replicated in every other cell.
There’s a caveat to all this, which we’ll come to later. But for now, this is all you need to know.
Now let’s look at each of the different chromosomal abnormalities in turn.
About numerical abnormalities (AKA “chromosomal aneuploidy”)
What are “aneuploid” chromosomes all about?
Chromosomal aneuploidy (try writing that while drunk) is when there’s any number other than 46 chromosomes in a cell. A cell with 45 chromosomes is aneuploid/abnormal – as is a cell with 47 or 48. And so on. Remember that if one cell in the human body is aneuploid, the other cells in the human body will also be aneuploid.
There are two common types of aneuploidy:
- Monosomy: when a chromosome in a particular pair is missing (giving a total of 45 chromosomes instead of 46)
- Trisomy: when there’s an extra copy of a chromosome – that is, there are three chromosomes instead of two (giving a total of 47 chromosomes instead of 46)
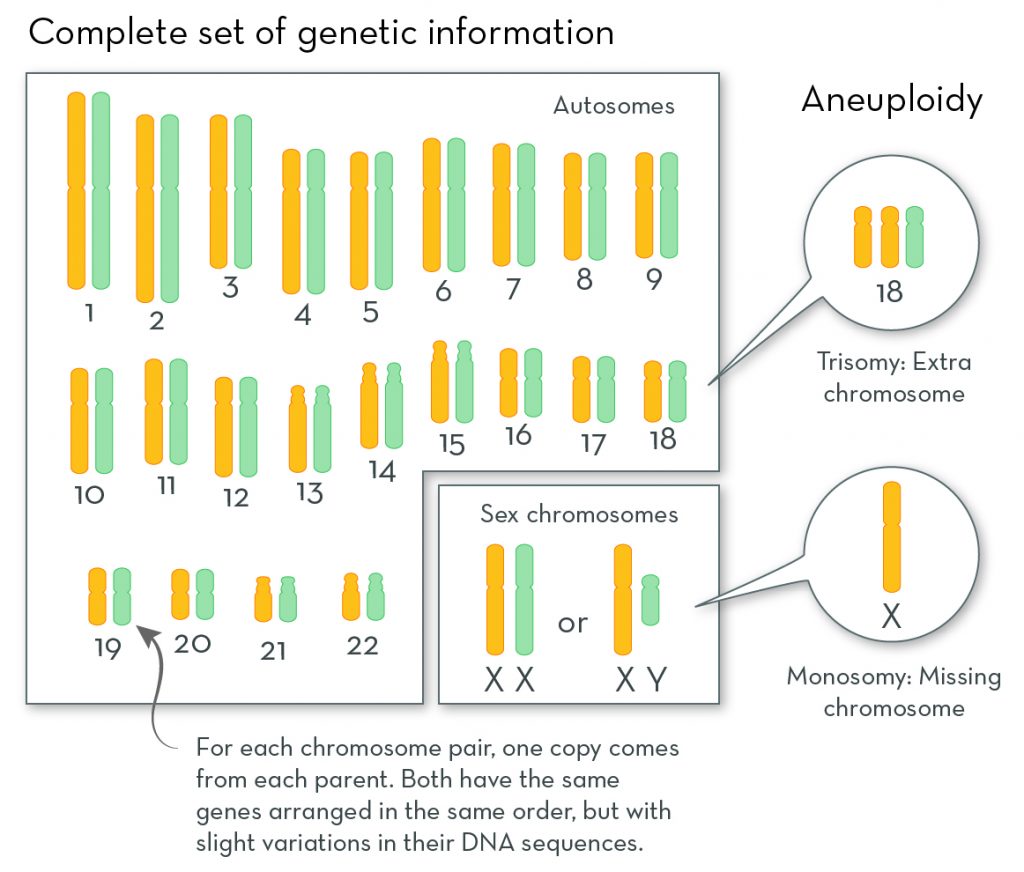
There are two other types, but they’re far less common – so they won’t get discussed much in this article:
- Nullisomy: when both chromosomes in a particular pair are missing (giving a total of 44 chromosomes instead of 46)
- Tetrasomy: when there are a total of four chromosomes rather than two in a pair (giving a total of 48 chromosomes instead of 46)
Whatever the abnormality, what you need to know is this: when there are too many or too few copies of a chromosome, the cell gets into all sorts of bother and confusion.
What are the risks associated with numerical abnormalities?
As mentioned above, aneuploidy is the leading cause of implantation failure and miscarriage. Some embryos do survive to term, but the baby will either die within a few days of birth or have a major developmental disability. When the abnormality is restricted to the sex chromosomes, the outlook is generally more positive.
Here are the most common numerical abnormalities that can survive pregnancy:
- Down syndrome (“trisomy 21”) – when there’s an extra chromosome in pair 21. According to one paper in the science journal Nature, “Individuals with DS all demonstrate signs of cognitive impairment, along with a more variable range of other symptoms, but patients routinely live to adulthood.”
- Patau syndrome (“trisomy 13”). According to the NHS website, trisomy 13, “in many cases, results in miscarriage, stillbirth or the baby dying shortly after birth”. It goes on to say that “More than 9 out of 10 children born with [Patau syndrome] die during the first year”.
- Edward syndrome (“trisomy 18”). The NHS says, “Sadly, most babies with [Edward syndrome] will die before or shortly after being born.”
- Embryos with extra sex chromosomes, such as Klinefelter syndrome – where males have two X chromsomes plus one Y chromosome or sometimes one X and two Ys (in addition to the 44 autosomes – making 47 chromosomes in total). Females have three Xs. Compared to most other numerical abnormalities, “… these sorts of sex chromosome trisomies are fairly benign. Affected individuals generally show reduced sexual development and fertility, but they often have normal life spans, and many of their symptoms can be treated by hormone supplementation.” (Source.)
- Females with a single copy of the X chromosome have a condition called Turner syndrome; their symptoms include growing up to be shorter than average, having underdeveloped ovaries, and certain visual characteristics. But while there’s no cure, many of the symptoms can be treated – and people with Turner syndrome can lead fairly normal lives.
- Monosomy 21. Babies rarely survive longer than a few weeks if the chromosome is completely missing, but they can survive is there’s only a “partial deletion” of a chromosome. Babies who survive (due to only a partial deletion) have symptoms similar to those of Down syndrome.
But remember: most numerical abnormalities don’t even survive pregnancy.
Here are some karyotype images showing different numerical chromosomal abnormalities:
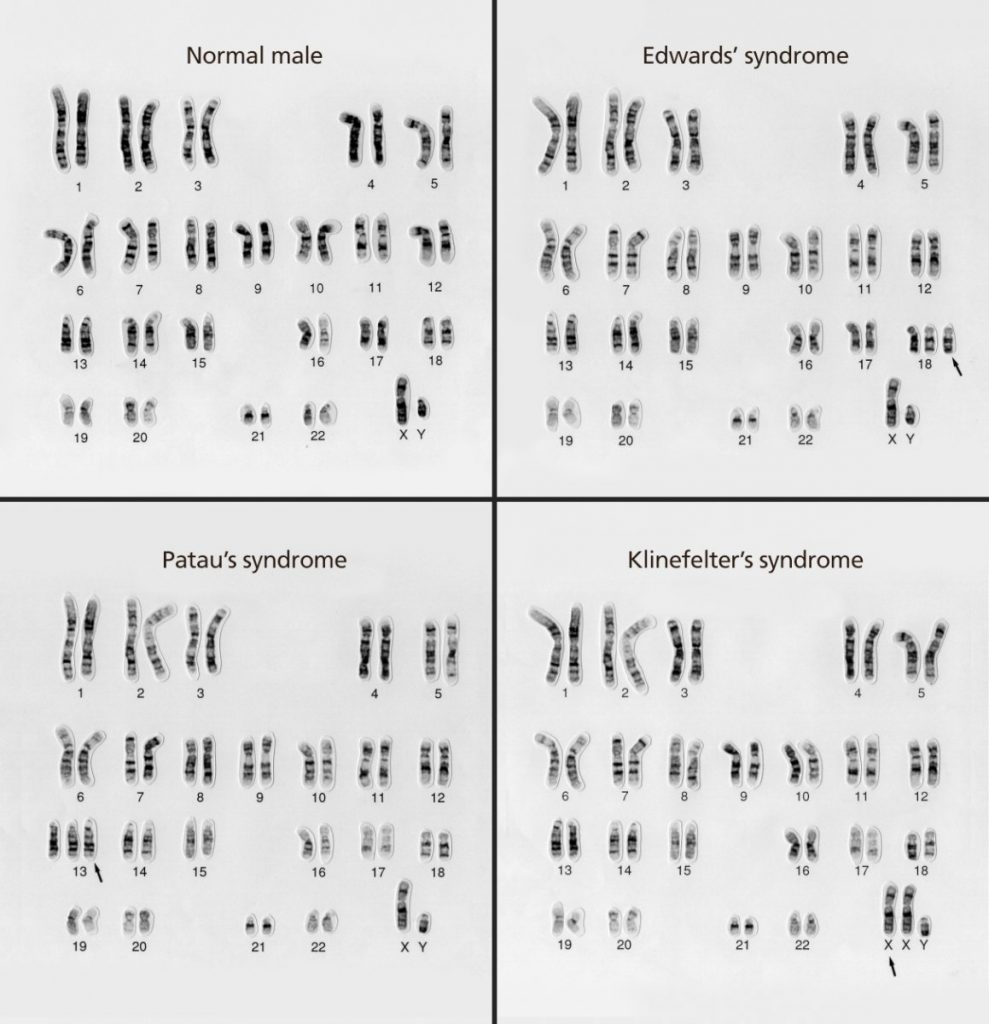
What causes aneuploid embryos?
There are two main ways in which embryo aneuploidy can happen:
Either…
- Soon after fertilisation, something can go wrong during cell division (mitosis) and the chromosomes won’t be distributed evenly. This cause doesn’t seem linked to maternal age.
Or (more likely – especially in older women)…
- The egg cell or sperm cell (but usually the egg cell) could contain an abnormal number of chromosomes. If this egg or sperm gets fertilised, the resulting embryo will have too many or too few chromosomes.
This particular problem relates to a process called meiosis, which will be covered in a lot of detail shortly.
Is aneuploidy ever an inherited condition? No. BUT structural abnormalities can be inherited, and the structural changes can lead to numerical changes. This will be covered later.
It’s important to remember that most of the time, the “original cause” is just bad luck. Yes: older women are more likely to have numerically abnormal eggs, but younger women have them too. And whether the original cause is an egg with the wrong number of chromosomes or an error in mitosis, it’s often just a case of Sod’s Law.
So remember: a healthy young woman with no family history of chromosomal abnormalities may still become pregnant with an abnormal embryo.

One embryo or two?
What are the risks and benefits of transferring more than one embryo? When clinics and hospitals recommend a single embryo transfer, do they have an underlying incentive?
What are the real facts?
I dug deep into all the research so you don't have to! Enter your email below for an easy-to-read, easy-to-understand guide to “one embryo vs two”.
I won't send you spam. Unsubscribe at any time.
Why are older women more likely to have egg cells with the incorrect number of chromosomes?
Before I attempt to answer this, it’s worth giving you some background information about how egg cells are formed in the first place.
So what you’ll now experience is a massive tangent in which I attempt to explain something called meiosis. Apparently, I learnt all this in GCSE biology many years ago. I have absolutely no recollection of that experience (something I’ll blame on the distracting cuteness of my teacher’s arse), so I’ve just spent about a fortnight’s worth of spare time trying to figure it out from scratch.
You don’t need to know how meiosis works, but it helps if you’re the kind of person who likes to understand everything about a topic. Meiosis isn’t a typo of “mitosis”, but it’s a similar(ish) process: both feature division and duplication, but in different ways from each other.
Tangent begins here
So, meiosis. Actually, let’s start (briefly) with mitosis. Mitosis, if you’ll remember, is when a cell continues to divide and duplicate. Mitosis occurs after an egg and sperm have fused together to form a zygote (a single cell with all 46 chromosomes). Mitosis works to replicate this single cell and its genetic material throughout the body.
Whereas mitosis occurs throughout the human body, meiosis is used for just one purpose: to produce sex cells (AKA eggs and sperm, or “gametes”).
As you should bloody well know by now, sex cells have half the usual number of chromosomes of a regular cell (that is, 23 instead of 46). It’s so that the egg and sperm can fuse together during fertilisation and plonk their respective 23 chromosomes together (in “homologous pairs”) – resulting in a full-set zygote of 46 chromosomes.
Meiosis is the process that makes it possible for the egg and sperm cells to have half the usual number of chromosomes. Without meiosis, egg cells and sperm cells would each contain 46 chromosomes. Without meiosis, therefore, humanity would have gone skewiff a LONG time ago.
But how does meiosis do this?
Unfortunately for all of us, the answer isn’t “magic”. The answer is, “It’s a process. And it involves two main stages. Actually, three.” (It’s all relevant… promise.)
Pull up a chair.
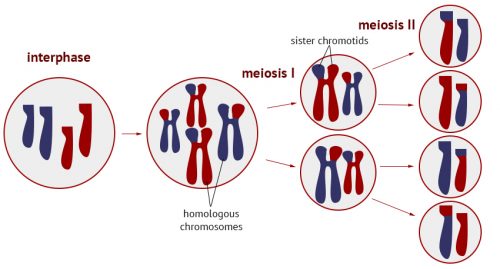
The interphase stage
“Interphase” happens before meiosis, and it happens in the reproductive organs. It’s when the chromosomes in a cell are replicated to create double the number of chromosomes – that is, 92 chromosomes in total. (Except it’s not really 92 chromosomes – and we’ll come to that shortly.)
The replicated chromosomes are connected to each other by something called a “centromere”, and these replicated chromosomes sit in their homologous pairs.
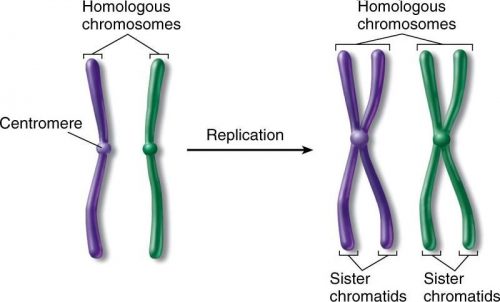
About the “chromatids” in the diagram: a chromatid is simply the name given to an individual chromosome that’s been newly copied.
In a “regular”, non-interphase-stage cell, you’d have 23 homologous pairs of unreplicated chromosomes. By now you’re familiar with this image:
In an interphase-stage cell, you’d have 23 pairs of replicated chromosomes. And it would look something like the image below. It’s the best image I could find, but it’s not great; hopefully you’ll get the gist.

So… there are now 92 chromosomes in a single cell. Except we don’t say there are 92 chromosomes because we count chromosomes according to how many centromeres there are.
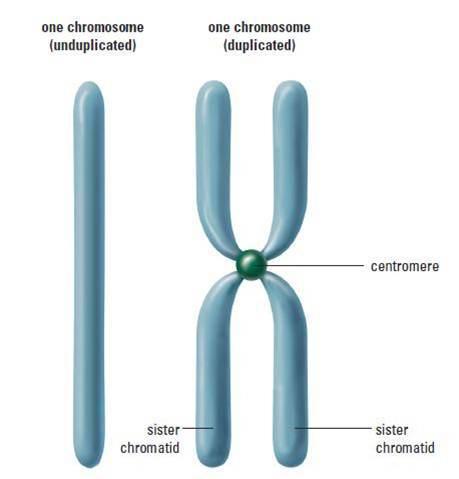
If you happen to be fussed about details, we now have 92 chromatids but only 46 centromeres – and the experts say that means we have 46 chromosomes.
Now we’re onto the official Stage 1 of meiosis.
Meiosis Stage 1
The replicated chromosomes line up in their pairs within the cell, and then they do this amazing thing where they swap bits of genetic information with each other. This is known as “crossing over”, and the resulting chromosomes are called “recombinant chromosomes”. The existence of recombinant chromosomes is one of the reasons why there’s such variety between offspring of the same parents.
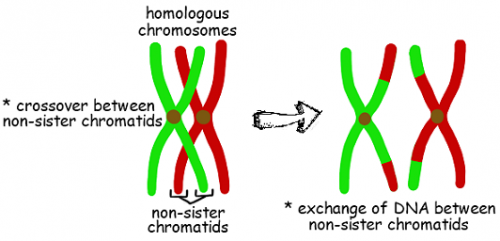
Super cute, huh?
Next up, these pairs of replicated, crossed-over (recombinant) chromosomes are pulled in opposite directions from each other, with each of the pairs on opposite sides of the cell:
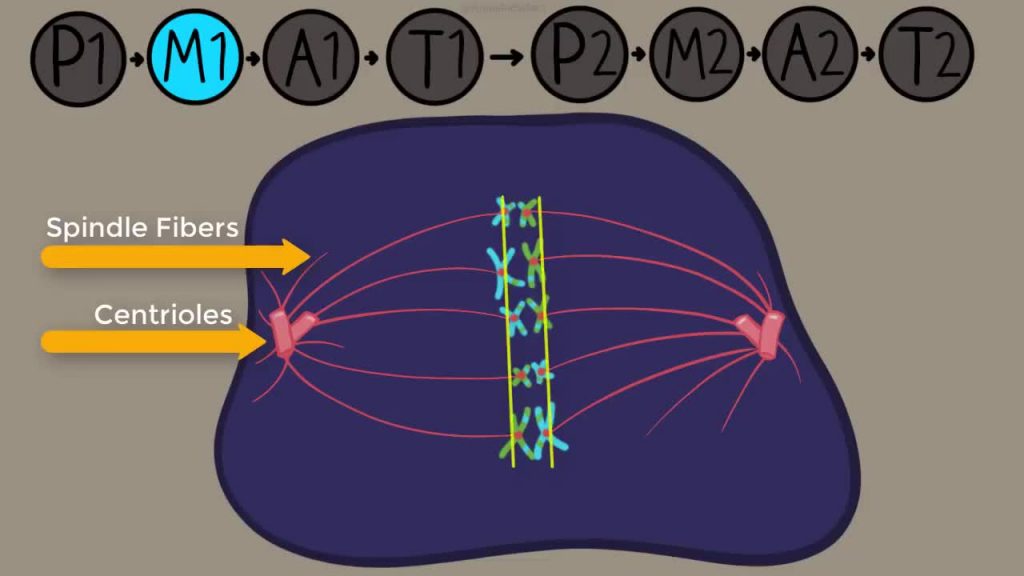
With thanks to the Amoeba Sisters YouTube channel: https://www.youtube.com/user/AmoebaSisters
The single cell reforms into two cells, each consisting of 23 replicated chromosomes. (23 chromosomes because – even though there are now 46 chromatids per cell rather than 92 – there are 23 centromeres. And remember: we count chromosomes according to how many centromeres there are.)
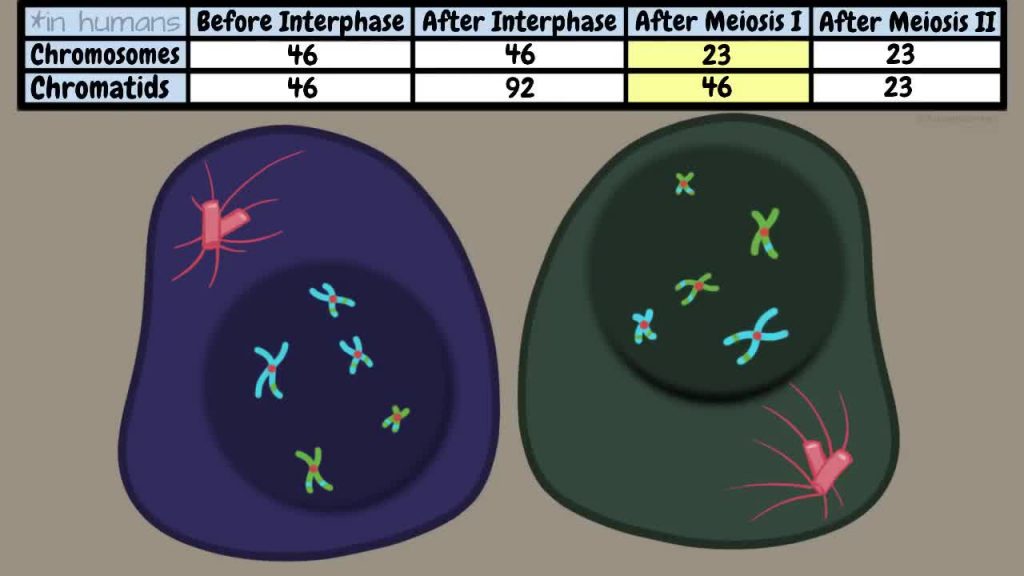
Meiosis Stage 2
You have your two cells, each with 23 chromosomes. In each cell the chromosomes line up again, but this time they’re in single file rather than pairs (their former “pair”, remember, is in the other cell).
Each chromosome is separated, but this time it separates at the centre – at the centromere – essentially separating the original chromatid from the “replicated” chromatid (the one that was created during interphase).
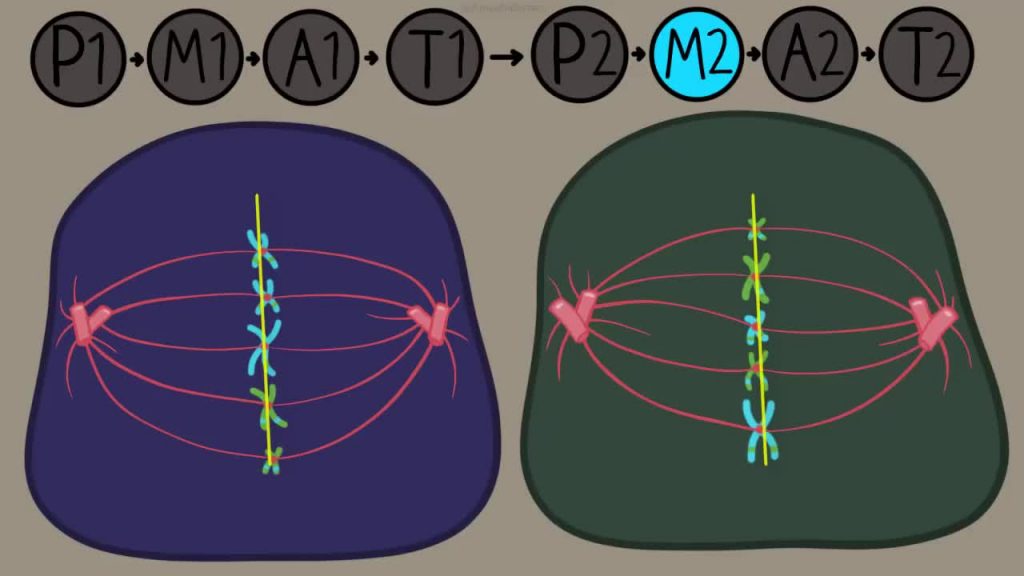
The chromatids move to opposite ends of their respective cells, and the two cells reform into four cells.
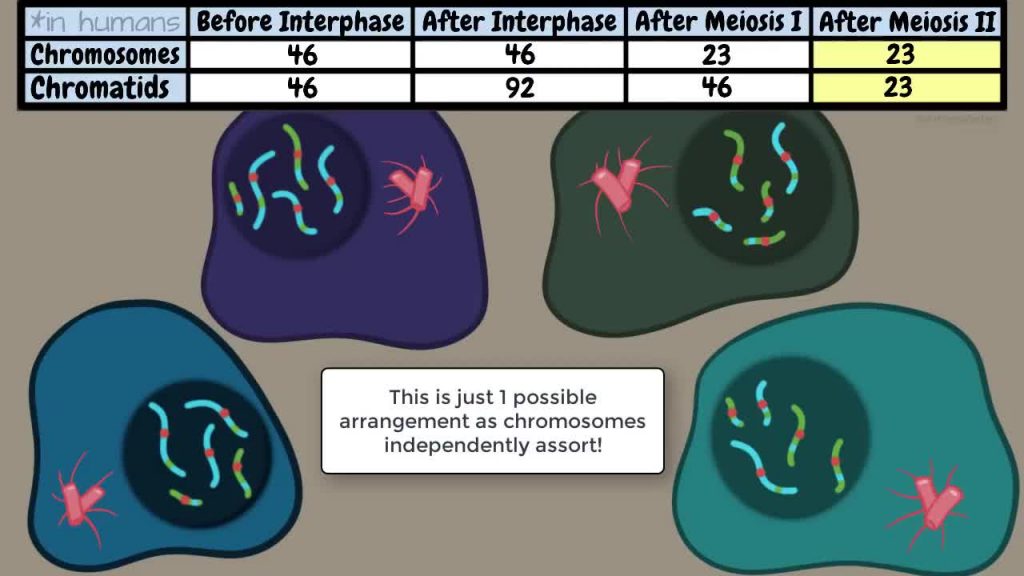
Each of the four cells is considered to have 23 chromosomes (rather than 11.5). Why? Because each chromosome retains a bit of centromere, and – as we now definitely know – chromosomes are counted according to how many centromeres there are.
So at the end of each meiosis process, we end up with four sex cells
Depending on whether they came from a male or a female, these sex cells are either four egg cells (i.e. four eggs) or four sperm cells (i.e. four sperm).
Here’s a lovely diagram to explain it all (showing just a few chromosomes to make it easier to understand):
Tangent ends here
There’s something I haven’t told you – and it’ll become super important later...
So you know meiosis Stage 1 and Stage 2? (If you didn’t read the explanation above, just nod your head and pretend here.)
In females, Stage 1 happens inside the eggs of a female foetus (!!!), which is impressively early and on-the-ball. (Women are born with all the eggs we’ll ever have: we don’t produce eggs as we go along.) Stage 2 happens when a particular developing egg is about to ovulate. That means the two stages of meiosis within one egg can easily be decades apart.
Back to our original question… Why are older women more likely to have egg cells with the incorrect number of chromosomes?
We (by which I mean, scientists) are pretty certain that potential chromosome-numbering problems occur during meiosis. If you haven’t read my massive tangent above, meiosis is the process by which egg cells (and sperm cells) are created.
No one really knows what happens during meiosis to cause chromosome-numbering problems, but one of the main theories these days is to do with proteins. The theory goes like this:
There are proteins present in chromosomes, which keep the chromatids of a chromosome nicely attached to each other. Lower levels of these proteins cause the chromatids to be more loosely connected.
When the chromatids are more loosely connected, they’re more likely to separate and “step out of line” before it’s required – i.e. before meiosis Stage 2 is fully underway. As a result, everything goes a bit wonky and the mature egg may end up with the wrong number of a particular chromosome. When the chromosomes don’t separate as they’re meant to, it’s called “nondisjunction”.
For example, the “nondisjunctioned” (not a word) egg might have two copies of Chromosome 21 as a result of not separating properly during meiosis. Once fused with a sperm cell, the resulting zygote may therefore end up with three copies of Chromosome 21, instead of the required two copies (one from the egg cell and one from the sperm cell).
What’s all this got to do with older women?
Well, older women have lower amounts of protein in their eggs, suggesting that as the eggs age, the levels of these proteins fall.
Caveat: this is all based on a study of mice, but scientists looked at 16 human eggs donated from women who’d done IVF to back up their research. They found that three eggs – all from women over 35 – were inclined to have the same “early separation” problem too. Other studies have backed up these findings.
It’s probably safer to assume that the protein theory is just one potential reason why age affects chromosomes in eggs – although it is, admittedly, the most popular reason at present. As another study puts it, while we don’t yet understand how age affects the likelihood of producing a normal egg, we can be fairly confident that there’s more than one direct way in which maternal age affects egg quality. (That is, it’s not just meiosis-related.) Here's more info on that study.
Can younger women have eggs with numerical abnormalities too?
Yes. All women have eggs with numerical abnormalities: it’s just that older women tend to have more eggs with numerical abnormalities – which means it’s more likely that a numerically abnormal egg will be fertilised.
About structural abnormalities
What makes a chromosome structurally abnormal?
Chromosomes contain strands of DNA in a highly organised way: there’s nothing random when it comes to the way in which the DNA is arranged or ordered (or there shouldn’t be, at least).
When large sections of DNA are either missing from or added to a chromosome, it’s referred to as a structural abnormality. There are a few different types, and they’re all caused by what are known as “mutations”. Mutations are changes in our genetic material. They’re often neutral and cause no bad effects, but some mutations are more harmful.
As mentioned earlier, structural abnormalities are far less common than numerical ones.
Here are the main types of mutation:
- Deletion: when part of a chromosome is missing. Deletions are also known as partial monosomies, and they can occur in any part of any chromosome. A person with a deletion only has one copy of a particular chromosome segment instead of the usual two. (The usual two copies come from each of the two homologous chromosomes.)
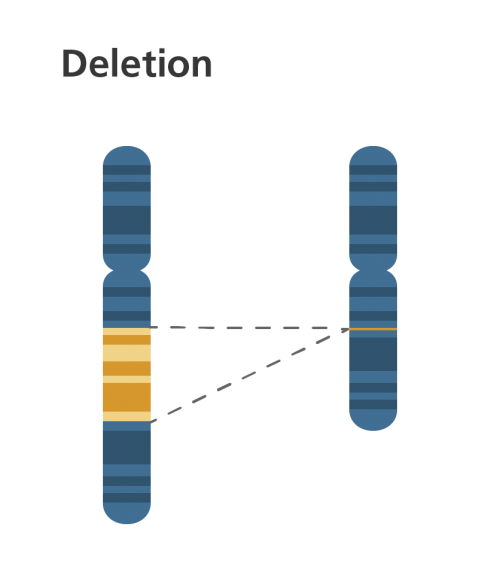
- Duplication: when part of the chromosome is repeated, which results in extra genetic material. Duplications are also known as partial trisomies.
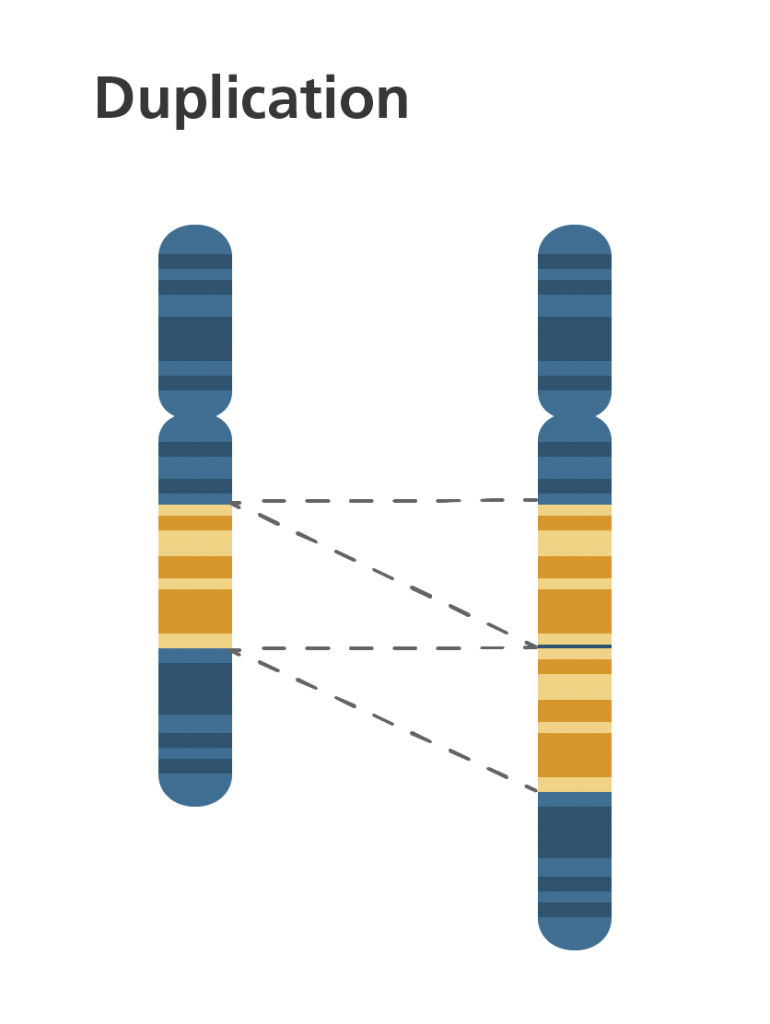
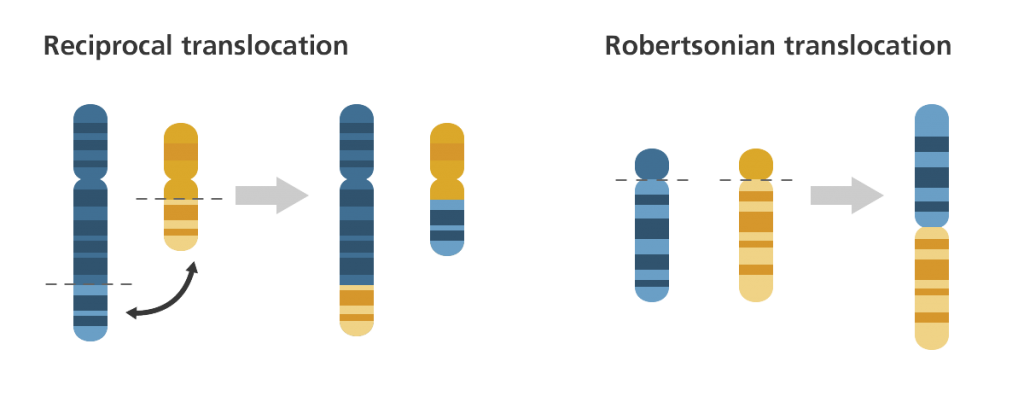
- Translocation: when chromosomes are rearranged in an unusual way. This is the most common form of structural abnormality. There are two main types:
Reciprocal: when segments from two chromosomes are exchanged.
Robertsonian: when an entire chromosome from one pair attaches to a non-homologous chromosome (i.e. from another pair), resulting in a total of 45 chromosomes instead of 46.
Note: these are both different from “crossing over” mentioned in meiosis earlier. Translocation occurs between non-homologous chromosomes (i.e. chromosomes from two different pairs), whereas crossing over occurs within one chromosomal pair. Crossing over is a normal process; translocation is not.
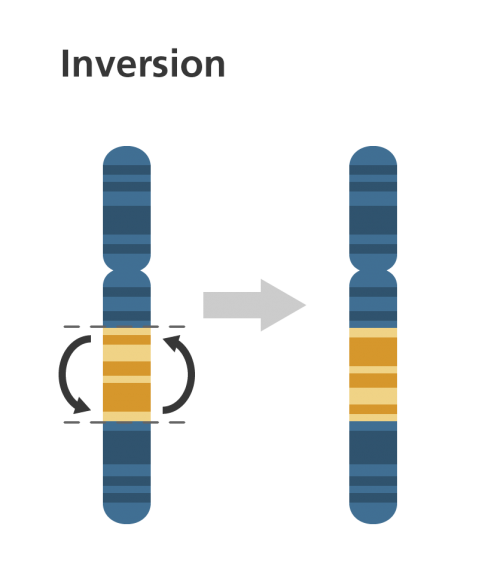
- Inversion: when a portion of a chromosome is inverted (in the opposite orientation).
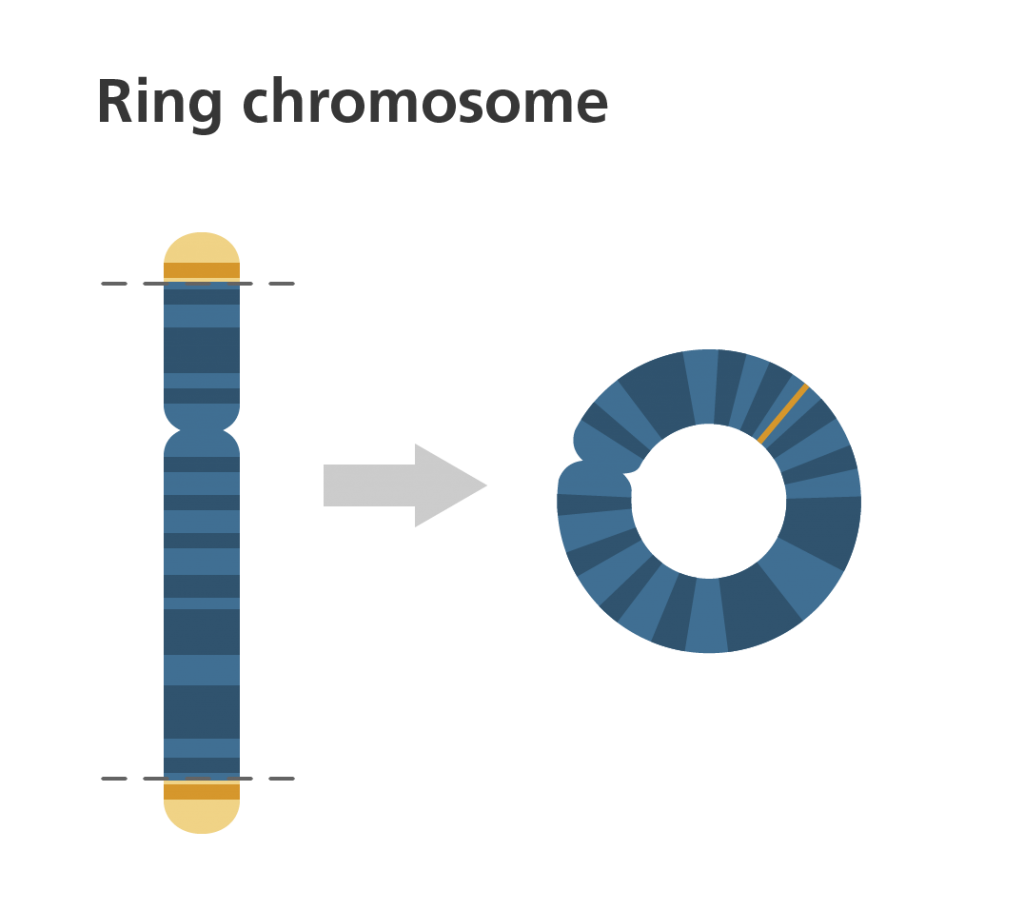
- Ring: when a portion of chromosome has broken off and formed a ring/circle shape.
All these structural abnormalities can be divided into two categories:
- Balanced: the complete chromosomal set is still present, even though it’s rearranged. Includes inverted and reciprocal translocated chromosomes, as well as some Robertsonian chromosomes. (Robertsonian chromosomes can be balanced if all the genetic information is still available – even if you have the wrong number of chromosomes).
- Unbalanced: there’s extra or missing information. Includes deletions, duplications, insertions, some Robertsonian translocations, and ring chromosomes.
Unbalanced abnormalities tend to be more serious, and balanced ones can actually go undetected sometimes. (The full set of DNA material is still present, so there may not be any symptoms.)
What kinds of problems do these structural abnormalities cause?
As with numerical abnormalities, it depends on the chromosome affected. Unlike numerical abnormalities, there are many structural abnormalities that don’t lead to miscarriage or infant death (although symptoms can still be severe and difficult to manage).
Below are some of the main types of structural abnormalities. I haven’t gone into detail on the symptoms, but I’ve provided links to more information on each one. Symptoms often involve distinctive facial/bodily characteristics and learning disabilities, among others.
- Wolf-Hirschhorn syndrome is caused by a deletion on the short arm of chromosome 4.
- Jacobsen syndrome is caused by a deletion on the long arm of chromosome 11.
- Cri-du-chat syndrome is caused by a deletion on the small arm of chromosome 5.
- 22q11.2 deletion syndrome is caused by a deletion of a small part of chromosome 22 near the middle of the chromosome at a location known as q11.2.
- In less than 5% of cases, Down syndrome is caused by a Robertsonian translocation of the long arm of chromosome 21 onto the long arm of chromosome 14. (Basically, the Robertsonian translocation causes a “trisomy 21” – which is a numerical abnormality.)
- XX male syndrome is caused by a translocation of a particular gene from the Y to the X chromosome.
- Ring chromosome 15 occurs when chromosome 15 breaks and the broken ends fuse together.

One embryo or two?
What are the risks and benefits of transferring more than one embryo? When clinics and hospitals recommend a single embryo transfer, do they have an underlying incentive?
What are the real facts?
I dug deep into all the research so you don't have to! Enter your email below for an easy-to-read, easy-to-understand guide to “one embryo vs two”.
I won't send you spam. Unsubscribe at any time.
What causes these structural abnormalities to occur?
As with numerical abnormalities, many structural abnormalities are a result of mistakes during the meiosis or mitosis stages. And (just as with numerical abnormalities) meiosis-related mistakes are more common – and “advanced maternal age” makes it more likely that these mistakes will occur.
But unlike numerical abnormalities, many structural abnormalities are also inherited (and not associated with maternal age).
To give one example of how an abnormality might be inherited… the chromosomes in your own body’s cells might be balanced reciprocal translocated chromosomes – that is, the segments from two chromosomes have broken off and swapped places with each other. You still have the same number of chromosomes as before and you still have all the genetic information you need – it’s just in a slightly different place compared to other people. You show no symptoms and live a normal, healthy life.
BUT because your chromosomes aren’t exactly where they’re meant to be, you’re considered a “carrier”. When you conceive, your embryo will only inherit half your chromosomes (the other half come from the father). If the father has “normal” chromosomes, there are four possible outcomes for any potential embryo:
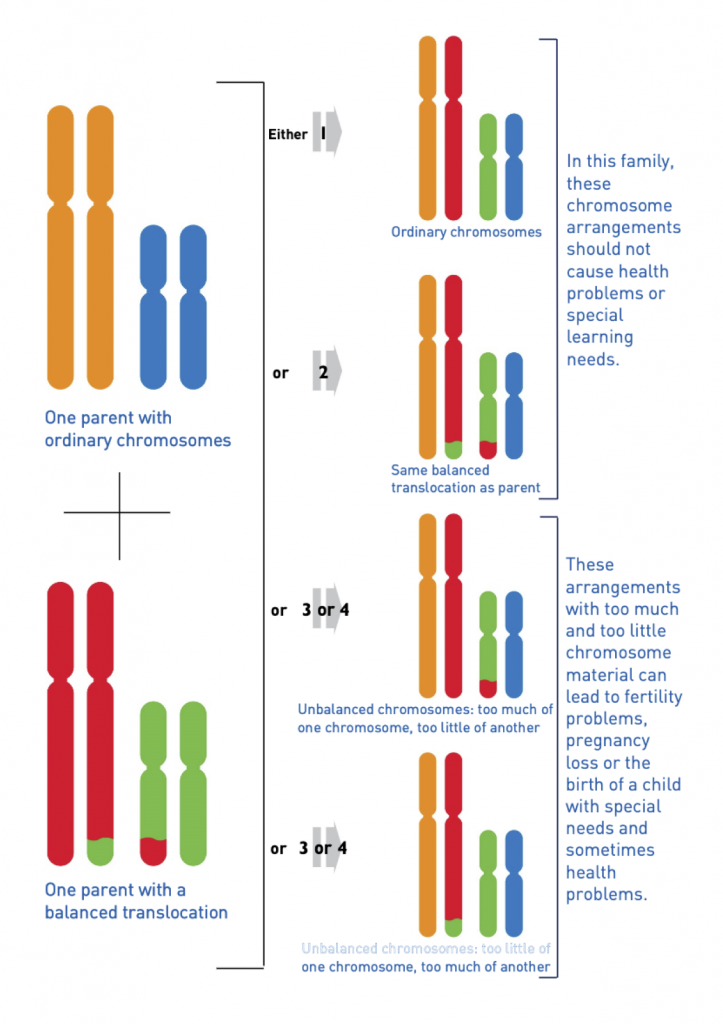
As described in the image above, a baby with unbalanced chromosomes has a high likelihood of being miscarried. If it survives pregnancy, it will have special needs and health problems.
It’s important to be aware that if you’re a balanced translocation carrier, it doesn’t mean there’s a one in four chance that embryos created by you and your partner will have unbalanced chromosomes. As one study found, “The incidence of abnormal pregnancies in carriers of balanced translocations depends strictly on the chromosomes involved in the translocations.”
Translocations are just one example of structural abnormalities that can either be inherited or a result of mitosis/meiosis.
How do I find out if I’m a carrier of inherited chromosomal abnormalities?
If you have problems getting pregnant or staying pregnant, it might indicate you’re a carrier – because the fertilised eggs aren’t surviving long. There are, of course, many other reasons why people struggle to get pregnant, so you’ll need to speak to your doctor about any tests and investigations you could have.
If you think you or your partner might be a carrier, you can get tested before trying for a baby: it’s just a simple blood test that involves examining some of your blood cells.
If you become pregnant with a baby and prenatal screening tests show that the baby has a chromosomal condition (like Down syndrome), it might indicate you’re a carrier – although there’s also a chance the condition is due to errors in meiosis or mitosis.
If I find out I’m a carrier, what are my options if I want to have a baby?
You could try to get pregnant the “regular” way, without any medical help. (Or if you need fertility treatment, you could try to conceive through IVF, ICSI, etc.) If/when you get pregnant, you could then decide whether or not to have a prenatal test to determine if your baby is likely to develop the chromosomal abnormality in question.
Note: this prenatal test will be different from the regular prenatal screening test that everyone is offered on the NHS.
Or you could have something called “preimplantation genetic diagnosis” (PGD) on your embryos. You’d need to do IVF, though: you couldn’t conceive naturally. More on PGD in a sec.
Or you might prefer to use healthy donor eggs or sperm instead. Adoption, of course, is always an option too.
About chromosomal abnormalities in sperm
Can sperm cells have numerical or structural abnormalities too?
Thank you for the reminder! Yes: a proportion of every man’s sperm will be aneuploid. But whereas women are born with all the eggs they’ll ever have (see my tangent on meiosis above for why this contributes to abnormalities), men produce new sperm continually. A 40-year-old man doesn’t have 40-year-old sperm. Instead he has – at most – three-month-old sperm.
But that doesn’t mean all men will have a similar proportion of euploid and aneuploid sperm. Some men will have a higher proportion of aneuploid sperm than others, and researchers have found it difficult to provide an “average” percentage figure.
As for what causes sperm aneuploidy… that’s a question for another article when I’ve done more research. One thing I discovered on my journeys across the internet, though (and which I may as well mention now), is that there might be a link between sperm aneuploidy and “teratozoospermia”.
Yup, that’s a real word – and you may be familiar with it if your partner has already been tested. Teratozoospermia is an increase in the percentage of abnormally shaped sperm in a sperm sample (anything above 4% is generally classified as teratozoospermia), and it’s one of the most common causes of male subfertility. Studies (like this one) suggest that men with teratozoospermia are more likely to have aneuploid sperm.
Other studies (like this one) link sperm aneuploidy with all types of male subfertility: oligozoospermia (low concentration of sperm in semen), asthenozoospermia (poor motility/ability to move around) and teratozoospermia (poor morphology).
When you read my article about preimplantation genetic screening (PGS), I discuss it mainly in the context of women who are older or have a history of miscarriage. But it might also be worth considering if your male partner is known to have subfertility issues.
The overwhelming number of embryo abnormalities, though, are due to errors from the female parent.
Can I find out if my embryos have any chromosomal abnormalities (numerical or structural)?
Yes: embryos can be tested before they’re selected for transfer into your uterus. (This is a big benefit of IVF over natural conception if you’re older or a carrier of a chromosomal abnormality.) There are two types of test:
- PGS (preimplantation genetic screening) tests for numerical abnormalities – which are the most common type of abnormality, and which disproportionately affect older women.
Numerical abnormalities generally aren’t inherited.
As mentioned earlier, it’s possible that an embryo could have a structural abnormality that isn’t inherited. PGS can pick up on some of these abnormalities too.
Read much more about PGS here.
- PGD (preimplantation genetic diagnosis) tests for structural abnormalities – which are less common, and which are often inherited. PGD is usually only offered to people who are known carriers of structural abnormalities.
I’ll have a new article about PGD available soon.
FYI PGS and PGD have recently been re-branded!
- PGS is now PGT-A, where “PGT” stands for “preimplantation genetic testing” and “A” stands for “aneuploidy”.
- PGD, meanwhile, has been divided into two new terms:
PGT-M (“monogenic”/single cell) looks for specific inherited conditions controlled by a single gene (like Huntington’s disease, sickle-cell disease and Fragile X syndrome) – a topic not covered by this article.
PGT-SR (“structural rearrangements”) looks for specific inherited chromosomal abnormalities (like translocations and inversions).
Next up: read all about PGS
Congratulations: I'm pretty certain you're in the top 1% of the population when it comes to understanding chromosomes!
Now go put that knowledge to good use: read up on how preimplantation genetic screening (PGS) works, find out the pros and cons, and reach a decision about whether you should shell out for it.
Read the article about PGS testing here.
(And I promise I'll write about PGD soon!)


