PGS TESTING IS A COMPLEX, NUANCED, AND MULTI-CAVEATED TOPIC, yet plenty of people act like it's good or bad for absolutely everyone.
When your fellow IVFers use Facebook groups and message boards to discuss whether or not to have PGS testing, their sentences should start with “If you have” or “If you want” – but most of the time all we see is “YES!!!” or “NOOOOO!!!”
And the “official” bodies aren't much better, by the way:
- Many private clinics tend to oversimplify to: “PGS is a great idea: go for it! Do you want to pay by credit card or bank transfer?”
- Meanwhile, the Human Fertilisation and Embryology Authority (the government regulator) is like: “RED LIGHT! RED LIGHT! STOP! Nooooooo!”
It’s hard to navigate through these two extremes in a way that’s balanced and fair, and also in a way that’s easy enough to understand – which is why it took me forever to write this article.
And finally, after many months of yelling at the internet to be less confusing while mainlining Pepsi Max and dreaming that all the world’s embryos are bitching about me, it's ready.
Has it been worth it? Let me know once you’ve read it.
Contents
Background information: chromosomal abnormalities
Before we start talking about PGS, we need to understand chromosomal abnormalities. Why? Because the purpose of PGS is to discover if any of your embryos have chromosomal abnormalities before they’re transferred into your uterus.
What is a chromosomal abnormality?
There are two types of chromosomal abnormality: structural and numerical:
- A numerical abnormality is an embryo that has the wrong number of chromosomes.
In sciencey speak, a numerically abnormal embryo is “aneuploid”, and a numerically normal embryo is “euploid”.
Numerical abnormalities are far more common than structural ones.
To discover numerical abnormalities, doctors use PGS (which stands for “preimplantation genetic screening”). - A structural abnormality is when large sections of DNA are either missing from or added to a chromosome.
Structural abnormalities are far less common.
To discover structural abnormalities, doctors use PGD (which stands for “preimplantation genetic diagnosis”).
The topic for this article is PGS testing, so we'll mainly be looking at numerical abnormalities. Soon I'll write about PGD and structural abnormalities.
Numerical abnormalities: what's meant by “the wrong number of chromosomes”?
Every cell in the human body should contain 23 pairs of chromosomes – making 46 chromosomes in total.
A numerical abnormality (AKA chromosomal aneuploidy… try writing that while drunk) is when there’s any number other than 23 pairs of chromosomes (46 in total) in a cell. A cell with 45 chromosomes is aneuploid/abnormal – as is a cell with 47 or 48. And so on.
If one cell in the human body is aneuploid, the other cells in the human body will also be aneuploid. (Actually, this isn't quite true – as will be explained in more detail later. For now, though, let's just assume it's true.)
Below is what you should see in each healthy male cell: 23 pairs of chromosomes. The first 22 pairs are numbered, and the final pair are “XX” or “XY” for female or male.
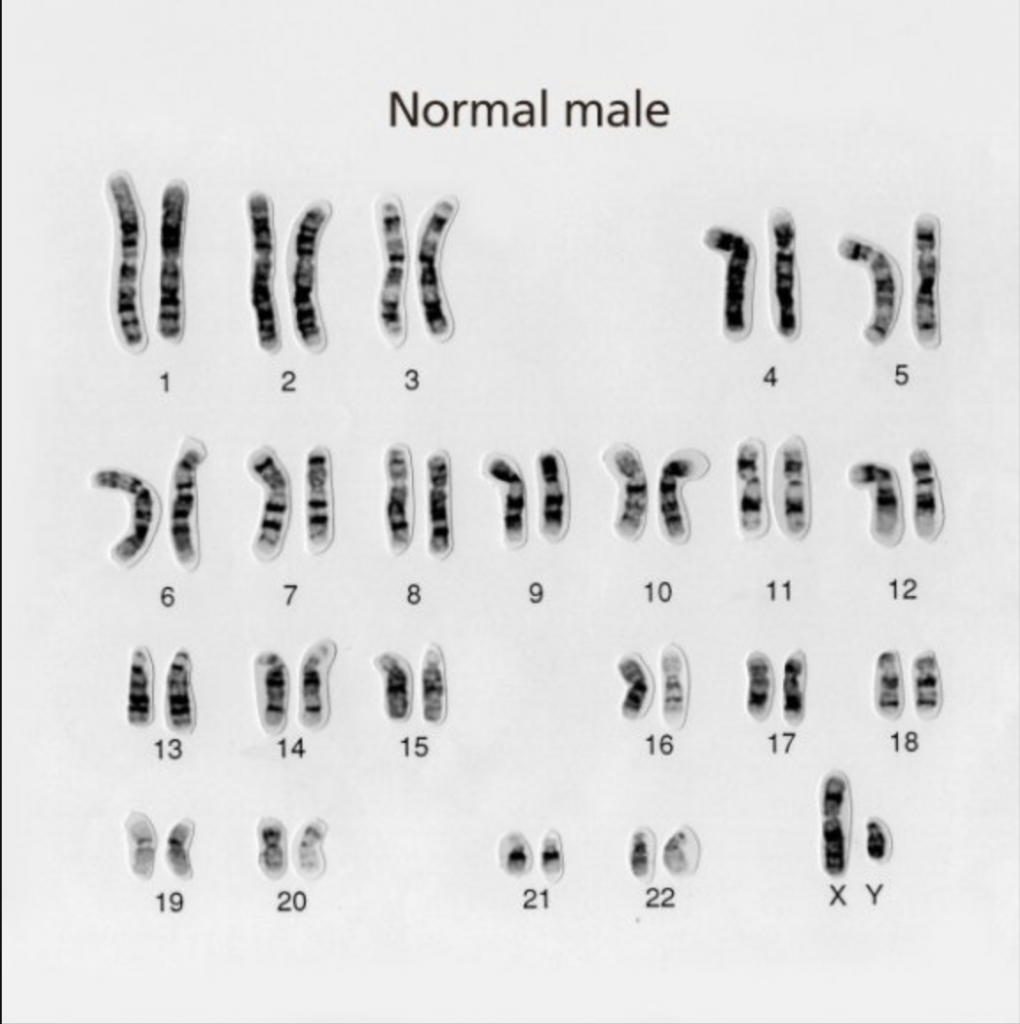
And here are examples of cells with chromosomal aneuploidy:
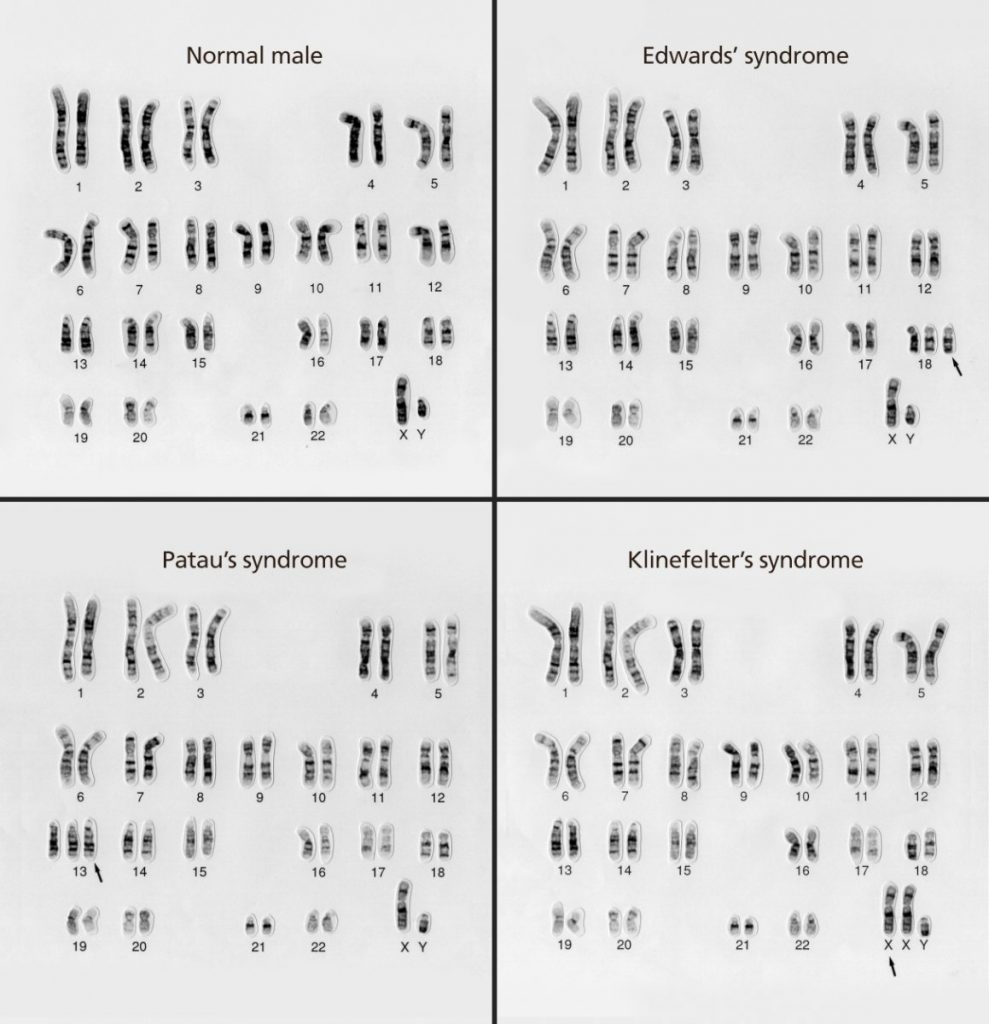
If you want to know much more about aneuploidy, cells, etc., I’ve prepared all my notes and background information for you to read here. It’s definitely worth reading them at some point, but I don’t want to digress too much from PGS testing in this article.
How common are numerical abnormalities in embryos?
Frighteningly common. According to one study, 42.9% of embryos (across all age ranges) are found to be aneuploid.
But chromosomal abnormalities increase dramatically with maternal age. Here’s some data from a study conducted in 2012 (note that the second column shows the percentage of normal embryos):
| Maternal age | % normal embryos |
| < 25 years | 70% |
| 25–29 years | 48% |
| 30–34 years | 42% |
| 35–39 years | 34% |
| > 39 years | 11% |
Igenomix (a reproductive genetics company) had similar results:
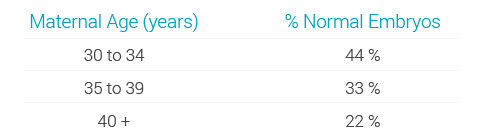
And this study from 2014 found that women under the age of 35 had a 60% chance of a blastocyst (an embryo five or six days after fertilisation) being euploid (having the right number of chromosomes), which reduced to 30% by the age of 41. The chance of getting NO euploids was about 10% for under 35s and about 50% by the age of 43. Other studies (like this one, but many others too) have the same sorts of figures.
Why do abnormalities become more common as women get older? Read this article for the answer.
What are the risks associated with numerical abnormalities?
Aneuploidy is the leading cause of implantation failure and miscarriage. Some aneuploid embryos survive to term, but the baby will either die within a few days of birth or have a major developmental disability. When the abnormality is restricted to the sex chromosomes, the outlook is generally more positive.
Here are the most common numerical abnormalities that can survive pregnancy:
- Down syndrome (“trisomy 21”). A trisomy is when there’s an extra copy of a particular chromosome – that is, there are three chromosomes instead of two.
Down syndrome is when there’s an extra chromosome in pair 21. According to one paper in the science journal Nature, “Individuals with DS all demonstrate signs of cognitive impairment, along with a more variable range of other symptoms, but patients routinely live to adulthood.” - Patau syndrome (“trisomy 13”). According to the NHS website, trisomy 13, “in many cases, results in miscarriage, stillbirth or the baby dying shortly after birth”. It goes on to say that “More than 9 out of 10 children born with [Patau syndrome] die during the first year”.
- Edward syndrome (“trisomy 18”). The NHS says, “Sadly, most babies with [Edward syndrome] will die before or shortly after being born.”
- Embryos with extra sex chromosomes, such as Klinefelter syndrome. Males with Klinefelter syndrome have two X chromosomes plus one Y chromosome or sometimes one X and two Ys. Females have three Xs. Compared to most other numerical abnormalities, “… these sorts of sex chromosome trisomies are fairly benign. Affected individuals generally show reduced sexual development and fertility, but they often have normal life spans, and many of their symptoms can be treated by hormone supplementation.” (Source.)
- Females with a single copy of the X chromosome have a condition called Turner syndrome; their symptoms include growing up to be shorter than average, having underdeveloped ovaries, and certain visual characteristics. But while there’s no cure, many of the symptoms can be treated – and people with Turner syndrome can lead fairly normal lives.
- Monosomy 21. A monosomy is when a chromosome in a particular pair is missing – that is, there’s one chromosome instead of two.
Babies rarely survive longer than a few weeks if the chromosome is completely missing, but they can survive is there’s only a “partial deletion” of a chromosome. Babies who survive (because of only a partial deletion) have symptoms similar to those of Down syndrome.
But remember: most numerical abnormalities don’t even survive pregnancy.
All about PGS testing
Brief recap: what does PGS have to do with numerical abnormalities?
PGS stands for “preimplantation genetic screening”, and it attempts to discover if any of your embryos have numerical abnormalities before they’re transferred into your uterus.
Errrrm… isn't PGS called something different these days…?
Well done, clever clogs.
Yes, that’s right: PGS and PGD have recently had a rebrand:
- PGS is now PGT-A, where “PGT” stands for “preimplantation genetic testing” and “A” stands for “aneuploidy”.
- PGD, meanwhile, has been divided into two new terms:
PGT-M (“monogenic”/single cell) looks for specific inherited conditions controlled by a single gene (like Huntington’s disease, sickle-cell disease and Fragile X syndrome).
PGT-SR (“structural rearrangements”) looks for specific inherited chromosomal abnormalities (like translocations and inversions).
This article covers PGS/PGT-A only. And because I hate change, I’m going to continue calling it PGS. (I’m almost certain I’m on the right side of history with this: remember “Choco Krispies”? “Consignia”? Exactly.)

Questions to ask your doctor at every stage of IVF treatment: free downloadable guide
IVF isn’t just overwhelming; it can also be a mind-boggling and sometimes terrifying experience because there’s so much to learn and so many rules to follow.
Problem is, doctors are busy – and they often don’t have the time to anticipate your concerns and provide all the information you might need.
This downloadable guide contains questions that will help you understand the process better, get the answers you deserve, and feel more in control of the situation (and your rights as a patient).
Enter your email address to receive it right away.
I won't send you spam. Unsubscribe at any time.
Who is PGS testing recommended for?
It depends who you ask. Some experts are against PGS in all circumstances (for reasons I’ll explain later), while others think it verges on essential for everyone. Unfortunately, these are the views you most often read about online.
But I'd say most experts fall into the third camp: “useful for certain types of people”. Those people include:
- Women who are aged 38 or older, because they’re more likely to have eggs with the wrong number of chromosomes. (Here's my explanation for why that's the case.)
- Couples who’ve had several miscarriages or failed IVF attempts without explanation.
- Men who have subfertility issues – because studies suggest that they’re also more likely to have a higher proportion of aneuploid sperm. (Here's my explanation why.)
How does PGS testing work?
PGS testing is normally performed on Day 5 embryos. By Day 5 of development (that is, five days after fertilisation), an embryo has divided into multiple cells – and PGS works by removing some of these cells for testing. Here’s what happens:
- A small hole is made in the shell of the embryo.
- Several cells are removed from the “trophectoderm” – which is the name given to the group of cells that will eventually form the placenta. (See diagram below.)
- These cells are then tested for numerical abnormalities.
- The results take about a week to come through, so all embryos are immediately frozen after PGS.
(Embryos implant in the uterus on or around day 7 or 8 of development, but if you wait a week for the results of PGS, the embryo would already be at least 12 days old. That’s why you need to freeze them after PGS, then wait for the results: freezing essentially stops them developing any further beyond day 5.) - Any embryos found to be aneuploid are usually discarded, while any euploid embryos are retained for future transfers.
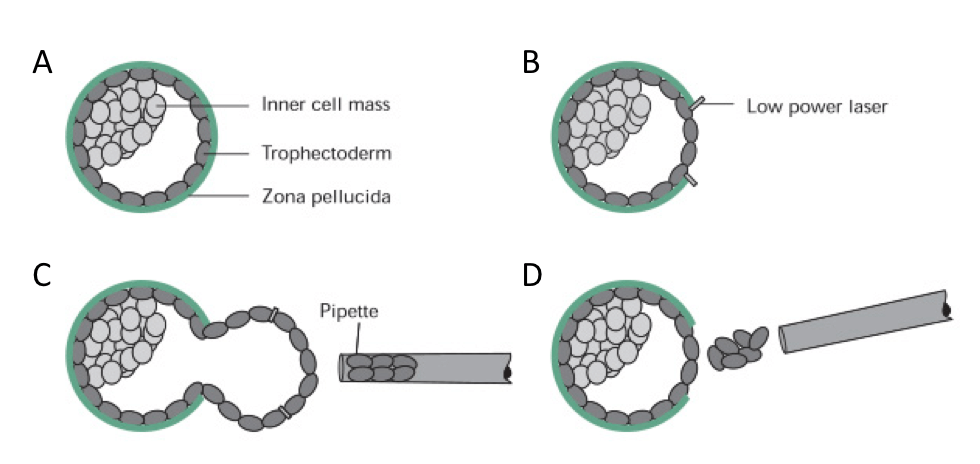
Things are a bit more complicated than I’ve just made out here, but that’s the gist.
Is PGS testing the same as embryo grading?
No. Embryo grading looks at the morphology (the visual appearance) of an embryo. PGS looks at the number of chromosomes in cells that have been extracted from the embryo. The vast vast majority of clinics will conduct embryo grading, whereas PGS is generally considered an “add on” treatment.
Read more about embryo grading here.
Ideally, you want an embryo that has a good grade AND the right number of chromosomes: that will give it the best chance possible for a successful implantation, pregnancy and birth.
Important:
- An embryo that has the best grade possible could still be aneuploid.
- A poorly graded embryo that’s chromosomally normal (“euploid”) has a greater chance of survival than a highly graded embryo that’s chromosomally abnormal (“aneuploid”).
How much does PGS testing cost?
PGS isn’t available on the NHS, so you’ll need to pay for private treatment if you want it. Prices vary from clinic to clinic. Here are some prices for PGS around the country:
- At CRGH in London, PGS costs from £2,850 (on top of the fees for IVF treatment).
- At CARE Fertility in Northampton, PGS costs £2,695 (on top of the fees for IVF treatment).
- At Oxford Fertility, PGS costs £2,300 (on top of the fees for IVF treatment).
- At Manchester Fertility, PGS costs £2,100 (on top of the fees for IVF treatment).
Note: each clinic will differ regarding how many embryos they’ll test for the price.
What’s the history of PGS testing?
OK, no one would actually ask this question. But I’ll pretend you did, because the answer could be important – as you’ll soon see.
PGS 1.0
In the 1990s, PGS was first conducted using a technique called FISH (fluorexcent in situ hybridisation) on Day 3 embryos – when there were barely any cells to biopsy. It used bad technology that could only screen a few chromosomes.
You’ll be unsurprised to learn that it didn’t take long for everyone to agree that PGS 1.0 didn’t seem to actually, errr, do anything useful. In fact, there’s a fair amount of evidence to suggest it had an adverse effect on pregnancy and birth rates. (Here’s one large-scale study.)
PGS 2.0
In the early 2000s, things moved on. With PGS 2.0, embryos were left until Day 5 (blastocyst stage) – when there are around 80 to 200+ cells to biopsy. About five of these cells were removed from the “trophectoderm” (the name given to the group of cells that will eventually form the placenta).
Unlike the old technology of PGS 1.0, ALL the chromosomes within these cells could now be examined – rather than just a few. (There were many different types of technology at this stage, including “SNP”, “aCGH” and “qPCR” – all of which fall under the umbrella of “PGS 2.0”. Don’t worry about what they all stand for: no one will test you.)
Everyone was delighted with PGS 2.0 until…
- They started to realise that people who were transferring embryos considered to be euploid were miscarrying. Those embryos later tested as aneuploid.
- They became concerned about the potential for “false positives”: embryos that could have good potential but were being discarded.
Brief interlude: the discovery of mosaicism
By the mid-2010s, embryologists discovered that blastocysts aren’t necessarily 100% euploid or 100% aneuploid: sometimes they’re a mixture. This is known as embryo “mosaicism” – a word that clearly wasn’t coined by someone with a lisp. (Earlier I’d said how if one cell in the body is euploid, all the others will be – and the same goes for aneuploid cells. Mosaicism shows how that's not always the case.)
Mosaic embryos are more likely to miscarry than euploid ones, but they stand a better chance of pregnancy and birth than aneuploid embryos. More on this in a sec.
If, therefore, you do PGS and it turns out you have no euploids but a few mosaics, most experts agree that it’s definitely better than having all aneuploid embryos (which almost always result in miscarriage or death soon after birth).
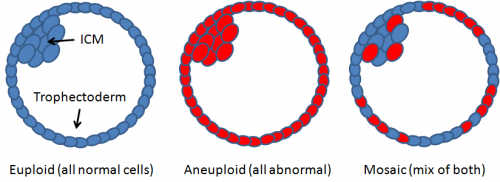
In the days of PGS 2.0, an embryo might be chromosomally mosaic, but the technology wouldn’t be able to pick up on it. (In fact, mosaicism wasn’t ever really discussed, because we barely knew about it.) As a result, an unidentified mosaic embryo would always end up in one of two crappy situations:
Either:
- The embryo would be considered euploid (because the mosaicism wasn’t detected), and it’d be transferred in place of a genuinely euploid embryo (because they’d both be considered equally euploid).
Or…
- The embryo would be considered aneuploid (because the mosaicism wasn’t detected), and it would be binned.
PGS 3.0
Technology has improved since PGS 2.0, and we’re now at PGS 3.0: it uses “NGS” (next generation sequencing), which can detect this mosaicism in embryos. Another benefit of NGS is that it’s better at detecting even partial deletions/duplications. (Partial deletions/duplications are considered structural abnormalities rather than numerical ones.)
So that’s where we are today: PGS 3.0 using a technology called NGS.
Except… this is the point at which they decided PGS needed a rebrand. Hence the new name: “PGT-A”. But screw that: I’m sticking with PGS.
A drawback of mosaicism detection in PGS 3.0…
For reasons I won’t bore you with, NGS can only detect mosaicism in excess of 20%. So if PGS results come back and an embryo has less than 20% aneuploidy, it will be considered euploid.
Likewise, if an embryo has more than 80% aneuploidy, it will be considered aneuploid. (This is something critics HATE about PGS, by the way: the classifications of euploidy and aneuploidy are entirely based on what the technology can detect, rather than what is actually viable as an embryo. We’ll come into this in more detail later.)
Where were we? Oh yes: 20% = euploid and 80% = aneuploid. In between the two extremes, we have varying degrees of mosaicism:
- < 20% aneuploid cells = euploid
- 21–40% aneuploid cells = low-level mosaic
- 41–80% aneuploid cells = high-level mosaic
- > 81% aneuploid cells = aneuploid
What are the success rates for PGS testing?
Most clinics state that PGS (3.0) results are correct 95% of the time. As you’ll have guessed, some studies agree with that number while others disagree. Regardless, that number alone can’t tell us whether PGS gives someone a better chance of getting pregnant and giving birth to a healthy baby.
And that’s because PGS success rates are more nuanced than they might initially seem.
Assuming PGS is 100% accurate in the following scenarios:
- If a woman has PGS and she’s told that all her embryos are aneuploid, that doesn’t mean PGS itself is “unsuccessful”. The outcome, though, is still the same: no chance of pregnancy, and no baby.
- If a woman transfers an embryo diagnosed as euploid after PGS but the pregnancy ends in miscarriage, that’s also not the fault of PGS: plenty of embryos miscarry or don’t implant, even when they’re chromosomally normal.
- If a woman transfers an embryo diagnosed as euploid after PGS and it does result in a healthy pregnancy and birth, that’s great – but, of course, that embryo could have been transferred without PGS and would also result in a healthy pregnancy.
Still… if PGS works as proponents say it works, we should be able to assume that when an embryo is transferred, a woman should be more likely to become pregnant and give birth to a healthy baby compared to non-PGS transfers. That’s because only euploids and mosaics will be transferred (and aneuploids will be discarded without “wasting” a transfer procedure on them).
So “births per embryo transferred” (the success rate measurement favoured by the HFEA) should be higher among those doing PGS testing.
That’s not the whole story, though. Of course it isn’t. Read on, because soon you’ll get to the all-important “pros and cons” lists.
What does “births per embryo transferred” mean?
“Births per embryo transferred” data shows the success rate for each individual embryo that’s transferred to the uterus, rather than the success rate for an IVF cycle as a whole (which could involve multiple transfer attempts with multiple different embryos from the same batch).
Does the data show that “births per embryo transferred” is higher among PGS patients, as we’d expect?
I don’t know.
Here’s the thing: women who do PGS testing currently tend to be older and/or have a history of miscarriages. For a fair and accurate study, we’d have to compare the exact same demographic for PGS vs non-PGS. It ends up being a small sample – perhaps too small to gauge anything from.
It’s for this reason that the HFEA doesn’t provide data on PGS success rates. (Remember that the HFEA doesn’t endorse PGS anyway, saying: “Until larger trials have been run and we have more evidence, there’s no guarantee that PGS can improve your chances of pregnancy.”)
As for individual clinics… they have a tendency to report vague, ill-defined success rates for PGS, but none of them will say the success rate per embryo transferred. (Perhaps it’s for the same reason as the HFEA: there are just too few cases of PGS to extrapolate from.)
If you’re happy with anecdotal information, here’s what I found on a few clinic websites:
- Glasgow Royal Fertility Clinic says:
“We are delighted in partnership with Genesis Genetics to be the first to offer this service to patients in Scotland. Patients are already seeing the benefit with a reduced time to pregnancy and a reduction in the risk of miscarriage.” - London Women’s Clinic says:
“Aneuploidy screening may improve pregnancy and live birth rates, because only those embryos with normal numbers of balanced chromosomes are selected for transfer. If all embryos are found to be aneuploid, screening will enable you to make an important decision about embryo transfer and IVF treatment.” - Bristol Centre for Reproductive Medicine (BCRM) says:
“Recent studies using… new [PGS] techniques showed that the chances of an embryo with a normal number of chromosomes producing a baby was more than 25% higher than those chosen based on the look of the embryo (morphology). While these results are extremely promising, they have not yet been replicated by other IVF centres. There needs to be further robust clinical and laboratory trials to assess whether or not PGS can significantly increase live birth rates.
“Although there are studies reporting improvements in IVF success rates using PGS, it is important to appreciate that there is other research suggesting that chromosome testing is of no benefit. There is also data suggesting that PGS may be of no benefit to patients with less than 3 embryos suitable for biopsy, although this is always a discussion the embryologist will have with you on the day of biopsy.”
So, nothing definitive from the clinics themselves either. Still, studies are ongoing, and many (including me) would argue that “success” includes finding out that all your embryos are aneuploid.
To summarise… what are the pros of PGS testing?
PGS testing can detect the presence of aneuploid chromosomes in embryos, which is a well-known cause of miscarriage and birth defects. By using PGS, it means the following:
- Increased chance of successful implantation. Healthy embryos are far more likely to implant and survive pregnancy and childbirth. By implanting an embryo without chromosomal abnormalities, you’ll increase the chance of IVF working – not just to pregnancy stage, but also to the stage of giving birth to a healthy baby.
- Success in less time (and money). Let’s say you have IVF treatment and end up with four embryos. Two of those embryos are aneuploid and two are euploid. If you don’t do PGS, you and your doctor won’t have this information available to you – so there’s a chance your doctor will transfer an aneuploid first, which results in miscarriage, and then another (this time frozen) aneuploid, which leads to another miscarriage.
And then you'll potentially run out of money/patience/sanity before you get round to transferring any of the euploids on ice. We don’t know how often this situation occurs, but we have to assume it’s more than zero.
If you have PGS first, your doctor will transfer one of the euploids instead.
Of course, it’s possible that the euploid embryos won’t lead to successful pregnancies/deliveries either, but they have a far, far better chance than aneuploid embryos. - Bad news will be delivered more swiftly. If all your embryos turn out to be aneuploid, you’ll avoid months – possibly years – of failed transfers.

Questions to ask your doctor at every stage of IVF treatment: free downloadable guide
IVF isn’t just overwhelming; it can also be a mind-boggling and sometimes terrifying experience because there’s so much to learn and so many rules to follow.
Problem is, doctors are busy – and they often don’t have the time to anticipate your concerns and provide all the information you might need.
This downloadable guide contains questions that will help you understand the process better, get the answers you deserve, and feel more in control of the situation (and your rights as a patient).
Enter your email address to receive it right away.
I won't send you spam. Unsubscribe at any time.
And what are the cons of PGS testing?
There are plenty of arguments against PGS, and it’s up to you to decide if they outweigh the pros…
Argument 1 against PGS: it’s expensive
Paying an extra £2,500 (ish) per cycle – on top of the £5,000 or so you’re already paying – is a LOT. Plenty of couples decide it’s better to put the money towards future cycles instead.
Argument 2 against PGS: you’ll have to freeze all your embryos
You have no choice about this: you’ll have to freeze all your embryos because there’s a delay in getting the results through. While many/most clinics now report frozen success rates on par with fresh ones, there’s still a risk that you may have a delicate euploid embryo that can’t survive the freeze-thaw process but would cope with a fresh transfer.
Plus it means more waiting around before you can do an embryo transfer.
Argument 3 against PGS: it gives patients a false sense of hope
Many patients aren’t really told what PGS attempts to achieve, and as a result they see it as something that will improve their chances of success.
PGS testing can improve a woman’s chances of faster success per egg retrieval procedure. (FYI When we say “per egg retrieval procedure”, it means “per batch of embryos created from one IVF cycle”. It could therefore mean “one embryo transferred” for one couple and “four embryos transferred – one after the other” for another couple.)
Why can PGS improve a woman’s chances of faster success per egg retrieval procedure? Because a chromosomally normal embryo will be selected for transfer first, whereas without PGS, no one knows if the embryo about to be transferred is euploid or not.
But if someone has zero euploid embryos after a cycle of IVF, PGS can only remove the time-wasting – not the heartache.
Not only that, but we need to remember that plenty of euploid embryo transfers don’t implant/lead to miscarriage too – either because of bad luck, or because something else was wrong with the embryo.
As the American Society for Reproductive Medicine (ASRM) says, “No single test… can accurately predict the risk of all defects in a child, and many birth defects, such as those related to environmental and toxic exposures and those that are random and unexplained, are not genetically based and may not be detected with genetic screening.”
Argument 4 against PGS: there’s a risk the embryo will be harmed during the biopsy
There’s currently no evidence (for blastocyst-stage embryos) that this is the case. But at least one expert has pointed out that studies claiming no ill effects to embryos are often based on biopsies of excellent-quality, robust embryos rather than more “delicate” embryos that might suffer more.
If you have just one or two embryos, you might decide it’s not worth the risk.
(Note: some believe that there’s a higher prevalence of aneuploidy in couples undergoing IVF or another assisted reproductive technology when compared to embryos conceived naturally. Rather than blaming PGS for causing the aneuploidy, they suggest that people undergoing IVF are more likely to have a higher chance of creating aneuploid embryos in the first place – and indeed that’s why they’re having trouble conceiving naturally.)
Argument 5 against PGS: the sample of cells is unrepresentative (and therefore inaccurate)
There are a couple of issues here:
- Issue 1
With PGS, usually five cells are biopsied from an embryo that contains anywhere from 80 to 200 cells.
Let’s say all five cells come back euploid. These five cells are supposed to represent the entire embryo, and – while there’s definitely a chance that the other 150-odd cells in the embryo are aneuploid – it’s more likely that the embryo as a whole is (as the biopsy would suggest) euploid.
When it comes to detecting levels of mosaicism, however, things get trickier. If, for example, one of those cells is aneuploid and the other four are euploid, the official diagnosis would be “low-level mosaicism”. Yet our biopsied sample might be unrepresentative, and we don’t really have any idea just how mosaic that embryo is. In fact, it could be the case that the other 150-ish cells are all perfectly euploid, and the one aneuploid cell is an outlier. Using the “20%” rule discussed earlier, that embryo should in fact be considered “euploid” and prioritised for transfer.
This isn’t just a theory. Many times, scientists have taken discarded mosaics and re-tested the entire embryo; they’ve found (again and again) that the original diagnosis was inaccurate. - Issue 2
The cells biopsied are taken from the trophectoderm (the cells that eventually form the placenta). But many (like these authors) wonder how accurately a trophectoderm biopsy represents euploidy/aneuploidy in the rest of the embryo.
Likewise, this article reports that “a trophectoderm biopsy specimen… might not necessarily be representative of the rest of the embryo in all instances… An important question is whether the proportion of abnormal cells in a mosaic biopsy specimen has any value for predicting the status of the remainder of the embryo.”
Argument 6 against PGS: the classifications are made up and arbitrary, and will therefore lead to throwing away perfectly good embryos
Some scientists don’t like the classifications given to different kinds of embryos with PGS 3.0. For example, Norbert Gleicher (who’s the President of the Foundation for Reproductive Medicine) points out that “The irrationality is… demonstrated by an embryo with 19 percent mosaicism now being allowed to be transferred as ‘normal’; yet an embryo with 21 percent aneuploidy load is considered ‘abnormal-mosaic’.”
Likewise, a letter to the editor of Reproductive BioMedicine Online by various obstetricians (including Norbert Gleicher) state that “The demarcation at the lower end (20% aneuploid DNA) was a purely technical one based on the minimum sensitivity level of [PGS]… the sensitivity limitations of a piece of laboratory equipment now officially determine embryo fate.” Read their entire letter for a particularly scathing review of PGS classifications and methods.
As PGS 3.0 classifications are – by most admissions – completely arbitrary, should we be holding it in such high regard whenever it's used? It could be argued that if PGS had never been conducted in the first place, we’d be none the wiser – and maybe that’s a better place to be.
Adding to the point above… some have found that once you have mosaic embryos on your hands, there’s barely any difference in pregnancy/birth rates – regardless of the extent of the mosaicism.
Argument 7 against PGS: there’s lots to suggest that mosaic embryos self-correct
Many specialists believe that mosaics are often able to self-correct their chromosomal abnormalities – although no one really knows why. (Various possible explanations have been suggested, but there’s nothing definitive.)
While the results of many studies imply that embryos diagnosed as mosaic have far worse prospects for survival (which in turn implies that only a certain amount of self-correction takes place), that doesn’t take away from the fact that many of them do seem able to self-correct.
Perhaps the low survival prospects among mosaics is because PGS is currently performed mainly on “high risk” embryos – that is, embryos from women who are older or who have a history of miscarriage.
Perhaps plenty of women not doing PGS have embryos that would be considered mosaic (even high-level mosaic), but they’re not being tested – and perhaps the “high risk” embryos have other reasons for miscarriage/implantation problems other than the abnormalities picked up by PGS.
Which leads to the question… if more women of all ages and risk profiles start opting for PGS in future, could we actually see pregnancy and birth rates decrease as a result – because women are being told their embryos are no good for transfer?
PGS seems to be becoming more and more popular, so we may well get our answer one day…
Argument 8 against PGS: if your clinic is using PGS 1.0 or PGS 2.0, steer well clear!
PGS 3.0 has its problems (see above!), but earlier technologies had far worse issues. Find out which type of PGS your clinic offers before making a decision about whether to go through with it.
So… is PGS testing worth it?
It depends. Many of those “cons” are only cons if you’re unaware of what PGS aims to achieve. If you decide to do PGS knowing full well what it’s for, and what it can and can’t do, you may well find it useful to have the information available to you.
But what about the fact that PGS testing might not be accurate?
Most studies suggest that PGS is very good at determining whether an embryo is euploid or aneuploid; it’s less good at figuring out levels of mosaicism. As embryos at all levels of mosaicism have been transferred successfully in the past, it seems to me that it makes the most sense to keep them all, rank them according to perceived mosaicism, and attempt to transfer them all (if necessary) over time.
As Michael Summers, Consultant at The Bridge Centre, says, the problems with accuracy in diagnosing mosaics mean we shouldn’t discard them after PGS testing. Instead, we should first order the embryos after conducting embryo grading, then shift “the rank order of embryos for transfer” after PGS, but keep hold of them all.
More and more clinics are following this model – but you’ll need to ask your own clinic what they do.
Anything else I should consider?
YES! A few random, unrelated points:
- Whether you do PGS or not, it’s a good idea to do a prenatal screening test once pregnant. Prenatal screening is a simple blood test combined with a scan, which tells you the odds that your baby has any of the three chromosomal disorders mentioned earlier that can survive pregnancy: Patau syndrome (“trisomy 13”), Down syndrome (“trisomy 21”) or Edward syndrome (“trisomy 18”).
(While the vast majority of embryos that test normal with PGS and implant will have a normal number of chromosomes, there’s still a very small chance that PGS gave you a “false negative”. That’s why it’s still recommended to get prenatal screening.)
If your screening test shows that there’s a high chance of a chromosomal abnormality, you can then – if you want to – have a more invasive test to get a definitive yes/no answer. - If you’ve read my article on embryo grading, you’ll know that it’s another method embryologists use to determine the health of an embryo.
As briefly discussed above, embryo grading is a largely subjective assessment of the “morphology” (the visual appearance) of embryos under a microscope. Certain visual characteristics imply that it has a better chance of implantation, pregnancy and birth.
Annoyingly, an embryo’s “grade” seems to have almost no correlation with whether it has chromosomal abnormalities or not. The very most we can say is that if your embryo grades aren’t top notch, it mayyyybe increases the risk that those embryos have chromosomal abnormalities too. But there are plenty of examples to the contrary.
Most embryologists agree that aneuploidy/euploidy is a far more important determining factor than an embryo’s grade. But we shouldn’t ignore embryo grading entirely – and I assume that embryologists would look at embryo grades AND PGS reports when deciding which embryo(s) to transfer.
Here’s a story about a doctor who believes that “If you have a good-looking embryo, forget the aneuploidy,” and has successfully transferred aneuploid (!) embryos with a good morphology. It’s just one anecdote, but it shows how non-cut-and-dried these things are. - Give some thought to your age (or your age when your eggs were collected). If you’re under the age of 40, your batch of embryos will contain about 40–50% euploids (ish) – so you might prefer to forego the PGS and try your luck. If you’re over the age of 40, your batch of embryos will contain about 10–20% euploids (ish) – so it might make more sense for you to do PGS. (There's more on all these figures in my article on chromosomal abnormalities.)
- Warning: controversial opinion (which isn’t MY opinion!) coming up! Dr Geoffrey Sher, a renowned endocrinologist in the US, points out:
“Given that about 50% of the eggs (and thus resulting embryos) of young women are euploid and competent, the transfer [of] up to 2 non-PGS tested blastocysts would likely yield the same results as would the transfer of a single PGS-tested euploid blastocyst. It follows that a patient/couple who are capable and willing to engage a twin pregnancy (which would occur in roughly 25% of such cases), might get as good a result by simply transferring two (2) untested blastocysts and in the process avoid the additional cost of PGS.”


